A new study published in Intelligent Oncology presents a deep learning–based system capable of accurately localizing renal tumors and classifying clear-cell renal cell carcinoma (ccRCC) using only non-contrast computed tomography (NCCT), offering a potential alternative to contrast-enhanced imaging in patients with contraindications to contrast media.
Background
Renal cell carcinoma (RCC) is a common urological malignancy, with ccRCC accounting for approximately 70%–80% of all cases. The current diagnostic standard relies on contrast-enhanced CT (CECT), which enables assessment of tumor vascularity and enhancement patterns. However, the use of iodinated contrast media carries risks of contrast-induced nephropathy and allergic reactions, limiting its use in patients with renal impairment or severe allergies. NCCT is widely available and safer, yet is traditionally considered insufficient for accurate tumor characterization.
Despite advances in artificial intelligence (AI), robust systems for automated ccRCC diagnosis directly from NCCT have remained lacking, representing a significant gap in the field.
Study Overview
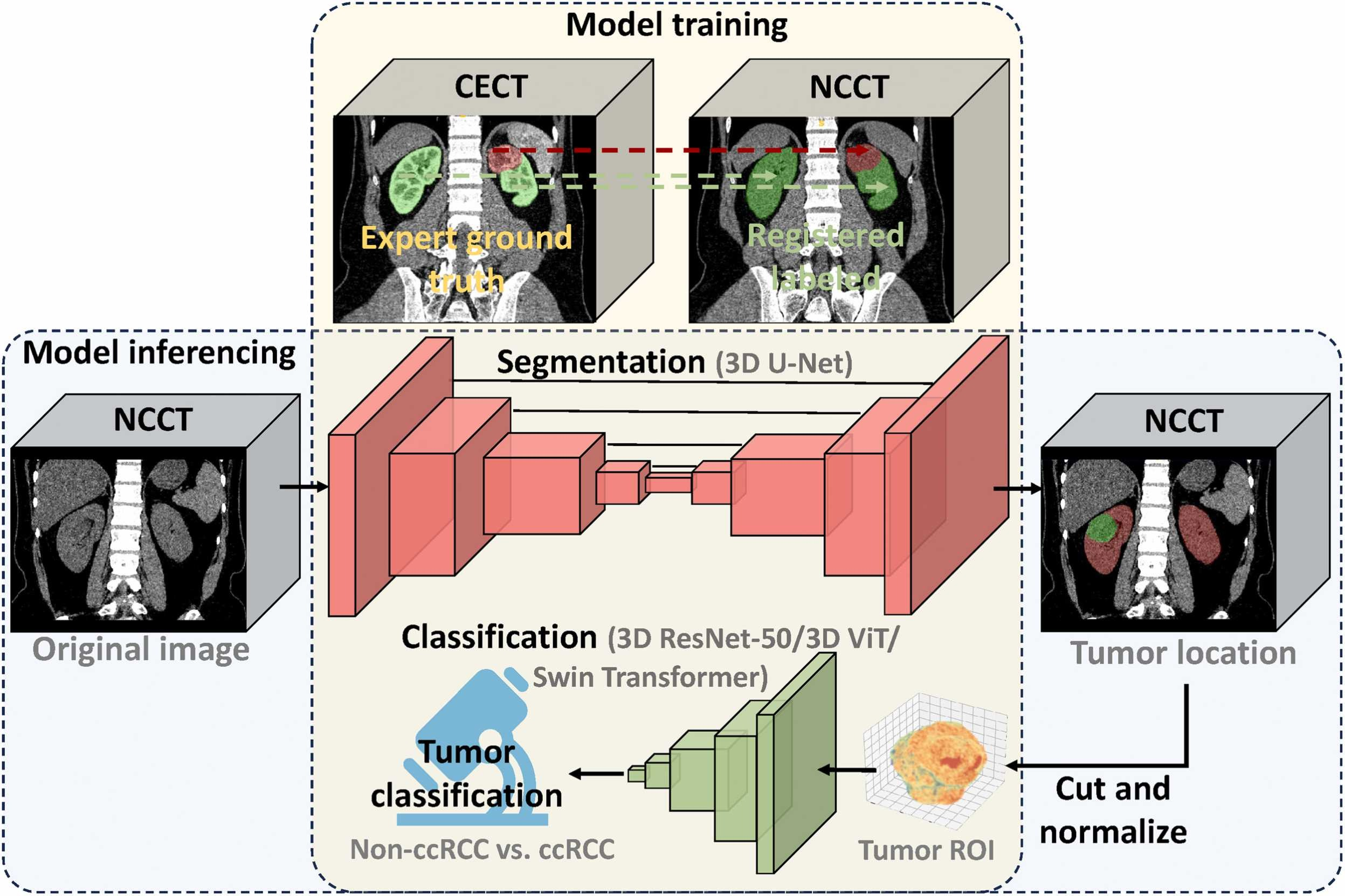
To address this gap, the investigators developed an integrated AI pipeline combining a 3D U-Net–based segmentation model with three classification architectures—3D ResNet-50, Vision Transformer (ViT), and Swin Transformer—to perform automated renal tumor localization and ccRCC classification using NCCT alone.
The multicenter retrospective study included 1,902 patients with paired NCCT and CECT scans from six independent medical centers. Expert annotations on CECT were propagated to NCCT using a 3D hybrid affine and deformable registration algorithm, with quality controlled by an automated kidney segmentation tool.
Three model configurations were developed and compared: an NCCT-only model, a CECT-only model, and a multiseries fusion model combining both series. Model performance was evaluated using internal and external test sets to assess robustness and generalizability.
Key Findings
The NCCT-based models demonstrated diagnostic performance non-inferior to that of CECT-based models:
NCCT segmentation achieved tumor Dice coefficients of 0.89 internally and 0.86 externally, approaching the performance of CECT-based models (0.96 internally, 0.94 externally).
NCCT classification models achieved AUCs of 0.76–0.82 on the external test set, with no statistically significant difference compared to CECT-based models.
The multiseries fusion model using the 3D ResNet-50 backbone achieved the highest performance, with an AUC of 0.88 on the external test set.
Simpler convolutional architectures (3D ResNet-50) consistently outperformed transformer-based models, suggesting that local texture features are particularly important for ccRCC classification.
The system operates fully automatically from raw NCCT to final prediction, integrating tumor segmentation and classification into a single pipeline.
Clinical Implications
The authors note that this AI-based approach has the potential to:
Enable renal cancer screening and diagnostic support for patients with contraindications to contrast media, such as those with renal impairment or severe allergies.
Serve as a triage tool for incidental renal masses identified on NCCT performed for other clinical indications, flagging lesions that warrant urgent urological evaluation.
Provide a contrast-free diagnostic pathway in resource-limited settings where contrast-enhanced imaging is less accessible.
By demonstrating that diagnostically relevant information can be reliably extracted from NCCT—a modality previously considered insufficient for such tasks—this work supports the integration of automated cancer screening into routine non-contrast examinations.
Conclusion
This study demonstrates that a deep learning system using only NCCT can localize renal tumors and classify ccRCC with accuracy comparable to models trained on CECT. The findings suggest that NCCT, a widely available and safe imaging method, can effectively screen for renal cancer without the need for contrast agents, with particular relevance for primary care settings and regions with limited resources.
Full article available on ScienceDirect:
https://www.sciencedirect.com/science/article/pii/S2950261626000130
Contact Information for Intelligent Oncology:
LinkedIn: @IntelligentOncology
X: @IntelligentOnco
Facebook: @intelligentoncology
Email Address: editorialoffice@intelligent-oncology.net
Official Website: https://www.sciencedirect.com/journal/intelligent-oncology
Submission Link: https://www2.cloud.editorialmanager.com/intonc/default2.aspx
